Image
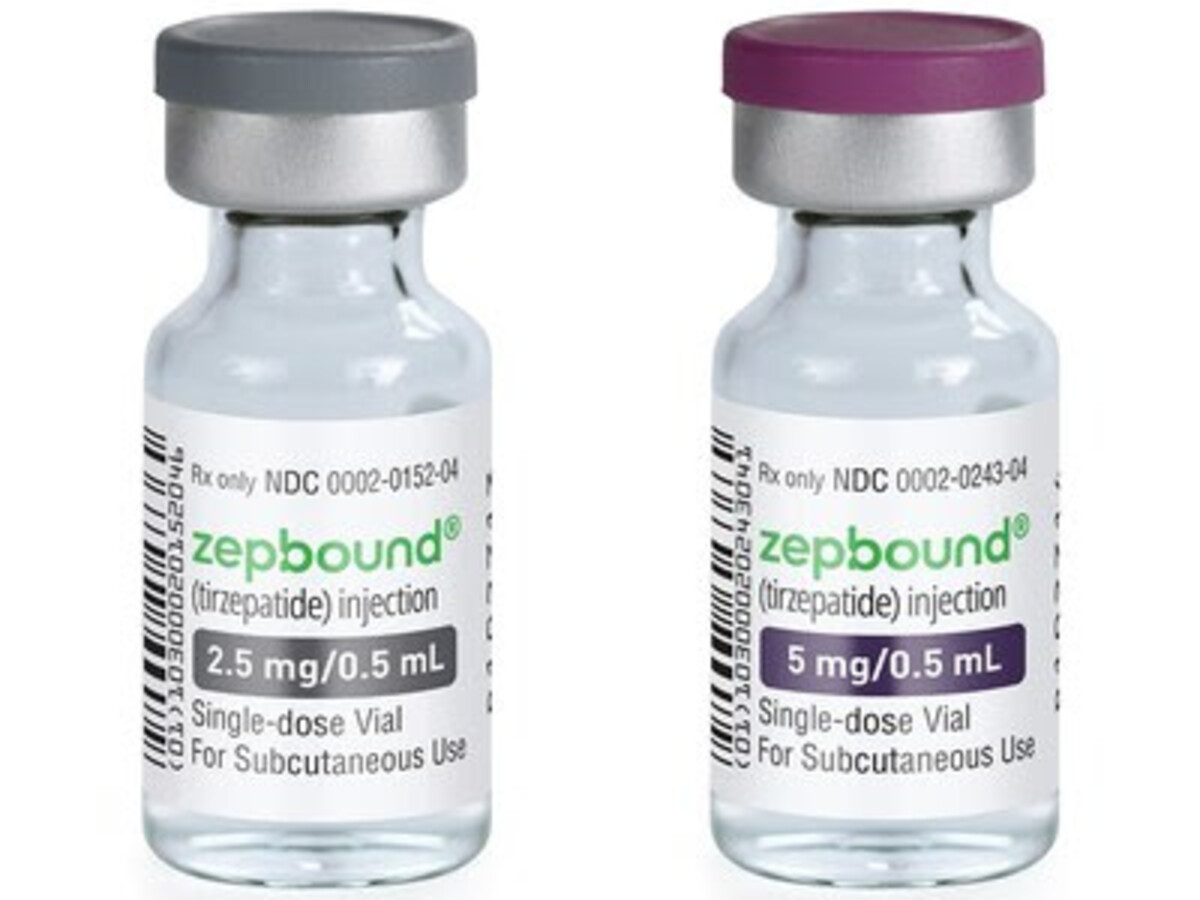

INDIANAPOLIS- Eli Lilly and Company (NYSE: LLY) today announced Zepbound® (tirzepatide) 2.5 mg and 5 mg single-dose vials are available for self-pay for patients with an on-label prescription, significantly expanding the supply of Zepbound in response to high demand. The single-dose vials are priced at a 50% or greater discount compared to the list price of all other incretin (GLP-1) medicines for obesity. This new option helps millions of adults with obesity access the medicine they need, including those not eligible for the Zepbound savings card program, those without employer coverage, and those who need to self-pay outside of insurance.
"We are excited to share that the Zepbound single-dose vials are now here, further delivering on our promise to increase supply of Zepbound in the U.S.," said Patrik Jonsson, executive vice president, and president of Lilly Cardiometabolic Health and Lilly USA. "These new vials not only help us meet the high demand for our obesity medicine, but also broaden access for patients seeking a safe and effective treatment option. In a clinical study, the 5 mg maintenance dose helped patients achieve an average of 15% weight loss after 72 weeks of treatment and has been a powerful tool for millions of people with obesity looking to lose weight and keep it off."
Lilly has created a new self-pay pharmacy component of LillyDirect where patients with a valid, on-label prescription from the health care provider of their choice can purchase the vials. Distributing the vials via this channel ensures patients and providers can trust they are receiving genuine Lilly medicine, building on the company's efforts to help protect the public from the dangers posed by the proliferation of counterfeit, fake, unsafe or untested knock-offs of Lilly's medications. Lilly has also taken a vocal stance against the use of obesity medicine for cosmetic weight loss; a multi-step verification process will help ensure the vials are dispensed only to patients who have a valid, on-label electronic prescription from their health care provider. Patients can also purchase ancillary supplies, like syringes and needles, and will have access to important patient-friendly instructional materials on correctly administering the medicine via needle and syringe.
"People living with obesity have long been denied access to the essential treatment and care needed to manage this serious chronic disease," said James Zervos, chief operating officer, Obesity Action Coalition. "Expanding coverage and affordability of treatments is vital to people living with obesity. We commend Lilly for their leadership in offering an innovative solution that brings us closer to making equitable care a reality. Now, it's time for policymakers, employers and insurers to work with pharmaceutical companies to ensure no one is left behind in receiving the care they deserve and need."
A four-week supply of the 2.5 mg Zepbound single-dose vial is $399 ($99.75 per vial), and a four-week supply of the 5 mg dose is $549 ($137.25 per vial) – less than half the list price of other incretin medicines for obesity and in line with the Zepbound savings program for non-covered individuals. The self-pay channel enables a transparent price by removing third-party supply chain entities and allowing patients to access savings directly outside of insurance.
"Despite obesity being recognized as a serious chronic illness with long-term consequences, it's often misclassified as a lifestyle choice, resulting in many employers and the federal government excluding medications like Zepbound from insurance coverage," said Jonsson. "Outdated policies and lack of coverage for obesity medications create an urgent need for more innovative solutions. Bringing Zepbound single-dose vials to patients will help more people living with obesity manage this chronic condition. We will also continue to advocate for a system that better aligns with the science."
In a clinical study, tirzepatide 5 mg, along with a reduced calorie diet and increased physical activity, achieved an average of 15% weight loss over 72 weeks compared to 3.1% for placebo. Zepbound is the first and only obesity treatment of its kind that activates both GIP (glucose-dependent insulinotropic polypeptide) and GLP-1 (glucagon-like peptide-1) hormone receptors. Zepbound tackles an underlying cause of excess weight. It reduces appetite and how much you eat. Zepbound is indicated for adults with obesity, or those who are overweight and also have weight-related medical problems, to lose weight and keep it off. Zepbound should be used with a reduced-calorie diet and increased physical activity. It should not be used in children under 18 years of age or with other tirzepatide-containing products or any GLP-1 receptor agonist medicines. Zepbound has not been studied in patients with a history of pancreatitis, or with severe gastrointestinal disease, including severe gastroparesis, and it is unknown if patients with a history of pancreatitis are at higher risk for developing pancreatitis on Zepbound.
Zepbound is also available in 2.5 mg, 5 mg, 7.5 mg, 10 mg, 12.5 mg, or 15 mg per 0.5 ml doses in a single-dose pen (autoinjector). The recommended maintenance dosages are 5 mg, 10 mg, or 15 mg injected subcutaneously once weekly.
To learn more about the Zepbound single-dose vial, please visit Zepbound.lilly.com.
To learn more about LillyDirect, please visit: LillyDirect.lilly.com.
INDICATION AND SAFETY SUMMARY WITH WARNINGS
Zepbound® (ZEHP-bownd) is an injectable prescription medicine that may help adults with obesity, or with excess weight (overweight) who also have weight-related medical problems, lose weight and keep it off. It should be used with a reduced-calorie diet and increased physical activity.
Warnings - Zepbound may cause tumors in the thyroid, including thyroid cancer. Watch for possible symptoms, such as a lump or swelling in the neck, hoarseness, trouble swallowing, or shortness of breath. If you have any of these symptoms, tell your healthcare provider.
Zepbound may cause serious side effects, including:
Severe stomach problems. Stomach problems, sometimes severe, have been reported in people who use Zepbound. Tell your healthcare provider if you have stomach problems that are severe or will not go away.
Kidney problems (kidney failure). Diarrhea, nausea, and vomiting may cause a loss of fluids (dehydration), which may cause kidney problems. It is important for you to drink fluids to help reduce your chance of dehydration.
Gallbladder problems. Gallbladder problems have happened in some people who use Zepbound. Tell your healthcare provider right away if you get symptoms of gallbladder problems, which may include pain in your upper stomach (abdomen), fever, yellowing of skin or eyes (jaundice), or clay-colored stools.
Inflammation of the pancreas (pancreatitis). Stop using Zepbound and call your healthcare provider right away if you have severe pain in your stomach area (abdomen) that will not go away, with or without vomiting. You may feel the pain from your abdomen to your back.
Serious allergic reactions. Stop using Zepbound and get medical help right away if you have any symptoms of a serious allergic reaction, including swelling of your face, lips, tongue or throat, problems breathing or swallowing, severe rash or itching, fainting or feeling dizzy, or very rapid heartbeat.
Low blood sugar (hypoglycemia). Your risk for getting low blood sugar may be higher if you use Zepbound with medicines that can cause low blood sugar, such as a sulfonylurea or insulin. Signs and symptoms of low blood sugar may include dizziness or light-headedness, sweating, confusion or drowsiness, headache, blurred vision, slurred speech, shakiness, fast heartbeat, anxiety, irritability, mood changes, hunger, weakness or feeling jittery.
Changes in vision in patients with type 2 diabetes. Tell your healthcare provider if you have changes in vision during treatment with Zepbound.
Depression or thoughts of suicide. You should pay attention to changes in your mood, behaviors, feelings or thoughts. Call your healthcare provider right away if you have any mental changes that are new, worse, or worry you.
Common side effects
The most common side effects of Zepbound include nausea, diarrhea, vomiting, constipation, stomach (abdominal) pain, indigestion, injection site reactions, feeling tired, allergic reactions, belching, hair loss, and heartburn. These are not all the possible side effects of Zepbound. Talk to your healthcare provider about any side effect that bothers you or doesn't go away.
Tell your healthcare provider if you have any side effects. You can report side effects at 1-800-FDA-1088 or www.fda.gov/medwatch.
Before using Zepbound
Review these questions with your healthcare provider:
❑ Do you have other medical conditions, including problems with your pancreas or kidneys, or severe problems with your stomach, such as slowed emptying of your stomach (gastroparesis) or problems digesting food?
❑ Do you take diabetes medicines, such as insulin or sulfonylureas?
❑ Do you have a history of diabetic retinopathy?
❑ Do you take any other prescription medicines or over-the-counter drugs, vitamins, or herbal supplements?
❑ Are you pregnant, plan to become pregnant, breastfeeding, or plan to breastfeed? Zepbound may harm your unborn baby. Tell your healthcare provider if you become pregnant while using Zepbound. It is not known if Zepbound passes into your breast milk. You should talk with your healthcare provider about the best way to feed your baby while using Zepbound.
How to take
Learn more
Zepbound is a prescription medicine. For more information, call 1-800-LillyRx (1-800-545-5979) or go to www.zepbound.lilly.com.
This summary provides basic information about Zepbound but does not include all information known about this medicine. Read the information that comes with your prescription each time your prescription is filled. This information does not take the place of talking with your healthcare provider. Be sure to talk to your healthcare provider about Zepbound and how to take it. Your healthcare provider is the best person to help you decide if Zepbound is right for you.
ZP CON CBS 08NOV2023
Zepbound® and its delivery device base are registered trademarks owned or licensed by Eli Lilly and Company, its subsidiaries, or affiliates.
LillyDirect® is a registered trademark owned or licensed by Eli Lilly and Company, its subsidiaries, or affiliates.
* The in-person and telehealth providers listed on the LillyDirect site are independent. Treatment decisions and prescribing practices are made based on the independent medical judgment of the provider's care team. They may prescribe medication or another course of treatment.
About Lilly
Lilly is a medicine company turning science into healing to make life better for people around the world. We've been pioneering life-changing discoveries for nearly 150 years, and today our medicines help more than 51 million people across the globe. Harnessing the power of biotechnology, chemistry and genetic medicine, our scientists are urgently advancing new discoveries to solve some of the world's most significant health challenges: redefining diabetes care; treating obesity and curtailing its most devastating long-term effects; advancing the fight against Alzheimer's disease; providing solutions to some of the most debilitating immune system disorders; and transforming the most difficult-to-treat cancers into manageable diseases. With each step toward a healthier world, we're motivated by one thing: making life better for millions more people. That includes delivering innovative clinical trials that reflect the diversity of our world and working to ensure our medicines are accessible and affordable. To learn more, visit Lilly.com and Lilly.com/news, or follow us on Facebook, Instagram and LinkedIn. P-LLY
PP-ZP-US-0986 08/2024 ©Lilly USA, LLC 2024. All rights reserved.
Cautionary Statement Regarding Forward-Looking Statements
This press release contains forward-looking statements (as that term is defined in the Private Securities Litigation Reform Act of 1995), including statements about the supply and access of Zepbound (tirzepatide) as a treatment for adults with obesity or overweight and reflects Lilly's current belief and expectations. However, as with any pharmaceutical product, there are substantial risks and uncertainties in the process of drug research, development, and commercialization. Among other things, there can be no guarantee that future study results will be consistent with the results to date, that Zepbound will receive additional regulatory approvals, that Zepbound will be commercially successful, or that Lilly will execute its strategy as planned. For further discussion of these and other risks and uncertainties, see Lilly's most recent Form 10-K and Form 10-Q filings with the United States Securities and Exchange Commission. Except as required by law, Lilly undertakes no duty to update forward-looking statements to reflect events after the date of this release.
SOURCE Eli Lilly and Company